The Role of the HPA Axis in Stress
The Role of the HPA Axis in Stress
Stress and HPA Axis
Scientifically, stress is defined as a state of real or perceived threat to homeostasis, or internal dynamic balance.1-4 Stress has many effects on the body and is closely linked to inflammation, activating inflammatory pathways in the brain and peripheral tissues.5 Chronic stress can result in permanent physiological and psychological changes and has been strongly associated with issues such as heart disease, cancer, skeletal fractures, liver disease, migraine headaches, anxiety, depression, metabolic syndrome, and type 2 diabetes.2,4,6 Stress-induced inflammation is likely a key factor in the development of these conditions.5
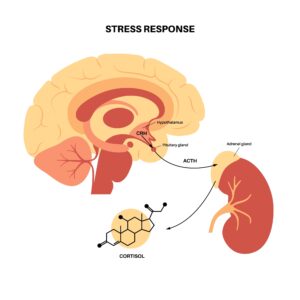
One major player in the stress response is the hypothalamic-pituitary-adrenal axis, or HPA axis. Together the hypothalamus, pituitary gland, and adrenal glands integrate many signals into a response to stressful events or stimuli. The hypothalamus is located in the brain and is the major command center of the body, maintaining homoeostasis and regulating complex functions such as emotion control and learning.7 The pituitary gland sits directly below the hypothalamus and acts like a thermostat, constantly assessing conditions to keep everything running well. It releases hormones to help the body function properly and regulates functions such as growth, metabolism, reproduction, and fluid balance. Similarly the adrenal glands, located on the kidneys, produce hormones that regulate major functions including metabolism, immune response, and blood pressure.
Actions of the HPA Axis
The HPA axis is composed of many pieces in the central nervous system (CNS) and peripheral tissues.1 The main players are localized in the paraventricular nucleus of the hypothalamus (PVN), the anterior lobe of the pituitary gland, and the adrenal gland.1 Neurons in the PVN synthesize and secrete corticotropin-releasing factor (CRF), which is a major regulator of the HPA axis.1
When there is a stressor present, CRF signals the anterior pituitary gland to release adrenocorticotropic hormone (ACTH) into circulation. Circulating ACTH targets the adrenal cortex, stimulating the synthesis and release of glucocorticoids (GCs), including cortisol, to regulate the stress response in peripheral tissues.3,6 When the stress response is activated, there are several behavioral and physiological changes that occur to improve the chance of survival, including increased awareness and cognition, increased respiratory rate and inhibition of feeding, digestion, growth, and reproduction.1
Glucocorticoids
Glucocorticoids are hormones that regulate physiological changes and downstream effects of the HPA axis in response to stress. They are important for homeostasis and normal functioning of vital systems that regulate growth, reproduction, immune response, behavior, and cellular metabolism.2,6 Glucocorticoids can also have a pro- or anti-inflammatory effect on the immune system, depending on the conditions.5 Under normal conditions, GC secretions follow a circadian rhythm, but a stressful event will override this and cause an immediate spike in GC levels.6
Cortisol
Cortisol, an important glucocorticoid, is secreted during acute and chronic stress.8 Under normal conditions, cortisol follows a pattern throughout the day where secretion increases during the night just before waking up with a short-lived spike following awakening, then declines steadily until sleep.4 This matches the energy demands of the body throughout the day (high during daytime and minimal during nighttime).6 Cortisol regulates the body’s response to stress, including triggering the release of glucose from the liver which can be used as a quick supply of energy. Dysregulated cortisol levels can have many detrimental effects on the body, including increasing abdominal obesity and decreasing bone mineral density.8
Regulation of HPA Axis
The HPA axis is tightly regulated through GCs as well as the endocrine, nervous, and immune systems.1 Glucocorticoids negatively regulate their own formation, suppressing the HPA axis in the hypothalamus and pituitary gland.1,4,6 In addition to regulation via glucocorticoids, the HPA axis follows the body’s natural circadian rhythm and is also regulated by brain stem neurons, appetite hormones including leptin from the hypothalamus, and the limbic system including the hippocampus, prefrontal cortex, and amygdala.1,4,6
The HPA Axis Coordinates the Stress Response
The HPA axis can be activated in response to acute or chronic stress and is activated by both external and internal stressors such as toxic foods, infections, or tissue damage.6
Acute stress
Short-term stress can actually be a good thing, helping to strengthen the body for future stress. The stress response begins quickly, within seconds, and seeks to help the body survive by redirecting oxygen and nutrients to the organs that are critical to survival including the heart and muscles.2 An acute stressor will induce a temporary spike in cortisol which can then regulate body systems needed to survive the stress. Cytokines can also activate the HPA axis through increasing GC receptor sensitivity which leads to increased GC concentrations.9
Chronic stress
While acute stress causes a sharp rise in cortisol secretion, long-term stress does not necessarily result in long-term elevated cortisol concentrations. Instead, it can result in lower-than-normal levels in the morning with higher-than-normal levels in the evening.4,8 One reason for this effect may be a protective mechanism by the HPA axis to avoid overexposure to stress hormones.4 Chronic stress can lead to exhaustion of several regulatory systems in the body including hormonal, cardiovascular, neural, and muscular systems.8 Long-term stress can also lead to structural changes in the brain, as well as metabolic impairments, hormonal and immune system changes, all of which contribute to chronic diseases.3
HPA Axis Dysfunction
HPA axis dysfunction can occur when any part of the system or its targets get off balance. Because the HPA axis is highly integrated with other networks and systems, the downstream consequences of altered HPA signaling can be significant. Hyper-activation of the HPA axis can lead to elevated GC levels which have been implicated in mechanisms underlying:1,3,4,6
- intestinal barrier dysfunction
- metabolic issues
- development of hepatic disorders
- mental health issues triggered by inflammatory responses
- autoimmune diseases
- hypertension
Cortisol imbalance
When chronic stress causes the HPA axis to be chronically activated or hyperactivated, cortisol levels remain high which results in many harmful downstream effects that further contribute to HPA axis dysfunction. For example, elevated cortisol can impair insulin binding to receptors, resulting in insulin resistance which increases oxidative stress and neurodegenerative changes, leading to greater HPA axis dysfunction.10 Hypercortisolism can also lead to disturbances in the immune system and metabolism.11
Alternatively, chronic stress and activation of HPA axis can damage the structure of the adrenal glands, resulting in chronically low cortisol levels (hypocortisolism) and impairing HPA axis function, which exacerbates the dysfunction.9
Inflammation
Inflammatory signaling molecules, including cytokines, are an important component of the stress response. However, in some cases inflammation can further exacerbate HPA axis dysfunction. For example, imbalance in the HPA axis can lead to increased fat surrounding internal organs which not only contributes to cardiometabolic disorders but also increases inflammation which worsens dysregulation of the HPA axis.12
Gut microbiota
Chronic stress can impair the barrier of the gastrointestinal tract, increasing gut permeability and triggering an inflammatory immune response.13 Dysbiosis of the gut microbiome can also contribute to an increase in cytokine levels and result in lower levels of short chain fatty acids (SCFAs).13 SCFAs are formed through the fermentation of indigestible carbohydrates and help maintain gut barrier integrity, down-regulate inflammatory processes, and can cross the blood brain barrier where they limit local inflammatory processes.13 SCFAs can also decrease the expression of genes that encode proteins involved in the HPA axis.13
Nutrition and Lifestyle in Regulating the HPA Axis
Healthy diet
Despite the urge to binge on comfort foods, choosing foods that are high in nutrients can help regulate the stress response. Consumption of high fat foods, often seen in the Standard American Diet, contributes to HPA axis dysregulation, even before weight gain begins.7 Specifically, the overabundance of saturated fatty acids can lead to fat deposition in the hypothalamus, leading to hypothalamic inflammation.7 The consumption of a high-fat diet can also lead to obesity which increases inflammation via pro-inflammatory cytokines, some of which can cross the blood brain barrier and affect the brain and HPA axis.7 Swapping saturated fats for unsaturated fatty acids may reduce hypothalamic inflammation.7 Probiotics and prebiotics can help restore stress-induced HPA axis dysfunction.13
Vitamins
Vitamin C is a very important nutrient in HPA axis balance. It is an antioxidant, immunomodulator, and involved in mediating the stress response.14 Vitamin C is particularly enriched in the adrenal glands and an increase in vitamin levels can enhance cortisol production and increase the anti-inflammatory effects of GCs.14 Additionally, ensuring adequate intake of B vitamins, vitamin A, vitamin D, and vitamin E, can help ensure a proper immune response, which is an important component of the HPA axis response.14
Minerals
Several minerals involved in brain health are important for HPA axis balance. Zinc is involved in many biochemical and physiological processes, including brain growth and function.15 Selenium, a component of selenoproteins, plays a role in antioxidant defense in the brain and nervous system.15 Finally, magnesium is heavily involved in the CNS, stress pathways, and oxidative pathways.15 Magnesium can alter the gut microbiota, influence oxidative and inflammatory responses, and reduce the release of ACTH, preventing hyperactivation of the HPA axis.15 Magnesium deficiency can lead to dysfunction in the CNS, including components involved in HPA axis pathways.15
Herbal support
Several herbs promote HPA axis balance. Ashwagandha (Withania somnifera), is a well-known adaptogen, meaning it promotes homeostasis of the body.16 Studies on the effects of ashwagandha on stress in humans have demonstrated promising results with reports of decreased stress and anxiety and improved sleep, quality of life, and well-being.16 Ashwagandha likely works through several mechanisms including decreasing HPA axis activation, inflammation, and oxidative stress.16 Ginseng (Panax ginseng) can also influence the HPA axis through its role as an antioxidant, reducing excessive inflammation, and supporting several aspects of brain health.17 Ginseng was able to alleviate HPA axis disorders including hypercortisolism in humans.17 The active ingredients of Licorice (Glycyrrhiza glabra) and Rehmanni (rehmannia glutinosa), glycyrrhizin and catalpol, respectively, have both demonstrated anti-inflammatory activities which supports a healthy stress response.18,19
Other lifestyle modifications
Beyond consuming a healthy diet and incorporating herbs and medicinal plants, developing healthy habits support HPA axis balance. Physical exercise can lower HPA axis activity, decrease oxidative stress and the pro-inflammatory immune response, and increase renal-adrenal function.20 Exercise may also be able to reduce diet-induced hypothalamic inflammation.7 Additionally, practicing meditation or mindfulness promotes relaxation and positively influences melatonin and serotonin levels.21 Finally, developing healthy sleep habits supports HPA axis function. Keeping a consistent sleep schedule helps maintain a normal circadian rhythm which can help support the natural rhythm of cortisol release and HPA axis activation.
The HPA axis is constantly working to maintain homeostasis in the body, responding to any stress that comes its way. Consuming nutrient-dense foods, utilizing herbs and medicinal plants, and engaging in regular physical activity are effective ways to support a healthy, well-regulated HPA axis and help maintain proper functioning of the body’s stress response.
- Smith, S.M., Vale, W.W. (2006). The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin Neurosci, 8(4):383.
- Kazakou, P., Nicolaides, N.C., Chrousos, G.P. (2022). Basic Concepts and Hormonal Regulators of the Stress System. Horm Res Paediatr, 1-9.
- Johnson, S. (2022). In Times of Adversity: A Neuroscience Perspective on Stress, Health, and Implications for Society Post-pandemic. Yale J Biol Med, 95(1):165.
- Murphy, F., Nasa, A., Cullinane, D., Raajakesary, K., Gazzaz, A., Sooknarine, V., Haines, M., Roman, E., Kelly, L., O’Neill, A., Cannon, M., Roddy, D.W. (2022). Childhood Trauma, the HPA Axis, and Psychiatric Illnesses: A Targeted Literature Synthesis. Front Psychiatry, 13:748372.
- Liu, Y., Wang, Y., Jiang, C. (2017). Inflammation: The Common Pathway of Stress-Related Diseases. Front Hum Neurosci, 11:316.
- Petrescu, A.D., Kain, J., Liere, V., Heavener, T., DeMorrow, S. (2018). Hypothalamus-Pituitary-Adrenal Dysfunction in Cholestatic Liver Disease. Front Endocrinol, 9:660.
- Dionysopoulou, S., Charmandari, E., Bargiota, A., Vlahos, N.F., Mastorakos, G., Valsamakis, G. (2021). The Role of Hypothalamic Inflammation in Diet-Induced Obesity and Its Association with Cognitive and Mood Disorders. Nutrients, 13:498.
- Cvijetic, S., Keser, I., Jurasović, J., Orct, T., Babić, Ž., Boschiero, D., Ilich, J.Z. (2022). Diurnal Salivary Cortisol in Relation to Body Composition and Heart Rate Variability in Young Adults. Front Endocrinol, 13:831831.
- Marino, L.O., Souza, H.P. (2020). Dysfunction of the hypothalamic-pituitary-adrenal axis in critical illness: a narrative review for emergency physicians. Eur J Emerg Med, 27(6):406.
- Habib, S., Sangaraju, S.L., Yepez, D., Grandes, X.A., Manjunatha, R.T. (2022). The Nexus Between Diabetes and Depression: A Narrative Review. Cureus, 14(6):e25611.
- Kokkinopoulou, I., Diakoumi, A., Moutsatsou, P. (2021). Glucocorticoid Receptor Signaling in Diabetes. Int J Mol Sci, 22(20):11173.
- Stapel, B., Jelinic, M., Drummond, G.R., Hartung, D., Kahl, K.G. (2022). Adipose Tissue Compartment, Inflammation, and Cardiovascular Risk in the Context of Depression. Front Psychiatry, 13:831358.
- Misiak, B., Loniewski, I., Marlicz, W., Frydecka, D., Szulc, A., Rudzki, L., Samochowiec, J. (2020). The HPA axis dysregulation in severe mental illness: Can we shift the blame to the gut microbiota? Prof Neuropsychopharmacol Biol Psychiatry, 102:109951.
- Toledano, J.M., Moreno-Fernandez, J., Puche-Juarez, M., Ochoa, J.J., Diaz-Castro, J. (2022). Implications of Vitamins in COVID-19 Prevention and Treatment through Immunomodulatory and Anti-Oxidative Mechanisms. Antioxidants, 11:5.
- Wang, J., Um, P., Dickerman, B.A., Liu, J. (2018). Zinc, Magnesium, Selenium and Depression: A Review of the Evidence, Potential Mechanisms and Implications. Nutrients, 10:584.
- Speers, A.B., Cabey, K.A., Soumyanath, A., Wright, K.M. (2021). Effects of Withania somnifera (Ashwagandha) on Stress and the Stress-Related Neuropsychiatric Disorders Anxiety, Depression, and Insomnia. Curr Neuropharmacol, 19(9):1468.
- Lee, S., Rhee, D. (2017). Effects of ginseng on stress-related depression, anxiety, and the hypothalamic-pituitary-adrenal axis. J Ginseng Res, 41:589.
- Wu, T., Khor, T., Saw, C.L.L., Loh, S.C., Chen, A.I., Lim, S.S., Park, J.H.Y., Cai, L., Kong, A.T. (2011). Anti-inflammatory/Anti-oxidative Stress Activities and Differential Regulation of Nrf2-Mediated Genes by Non-Polar Fractions of Tea Chrysanthemum zawadskii and Licorice Glycyrrhiza uralensis. AAPS J, 13(1):1.
- You, L., Peng, H., Liu, J., Cai, M., Wu, H., Zhang, Z., Bai, J., Yao, Y., Dong, X., Yin, X., Ni, J. (2021). Catalpol Protects ARPE-19 Cells against Oxidative Stress via Activation of the Keap1/Nrf2/ARE Pathway. Cells, 10:2635.
- Daniela, M., Catalina, L., Ilie, O., Paula, M., Daniel-Andrei, I., Ioana, B. (2022). Effects of Exercise Training on the Autonomic Nervous System with a Focus on Anti-Inflammatory and Antioxidant Effects. Antioxidants, 11:350.
- Gustafson, C. (2016). Herbal Support for the HPA Axis. Integr Med, 15(6):42.