Inflammaging and Whole Food Nutrition
How phytonutrients in whole foods can calm inflammation that contributes to age-related conditions
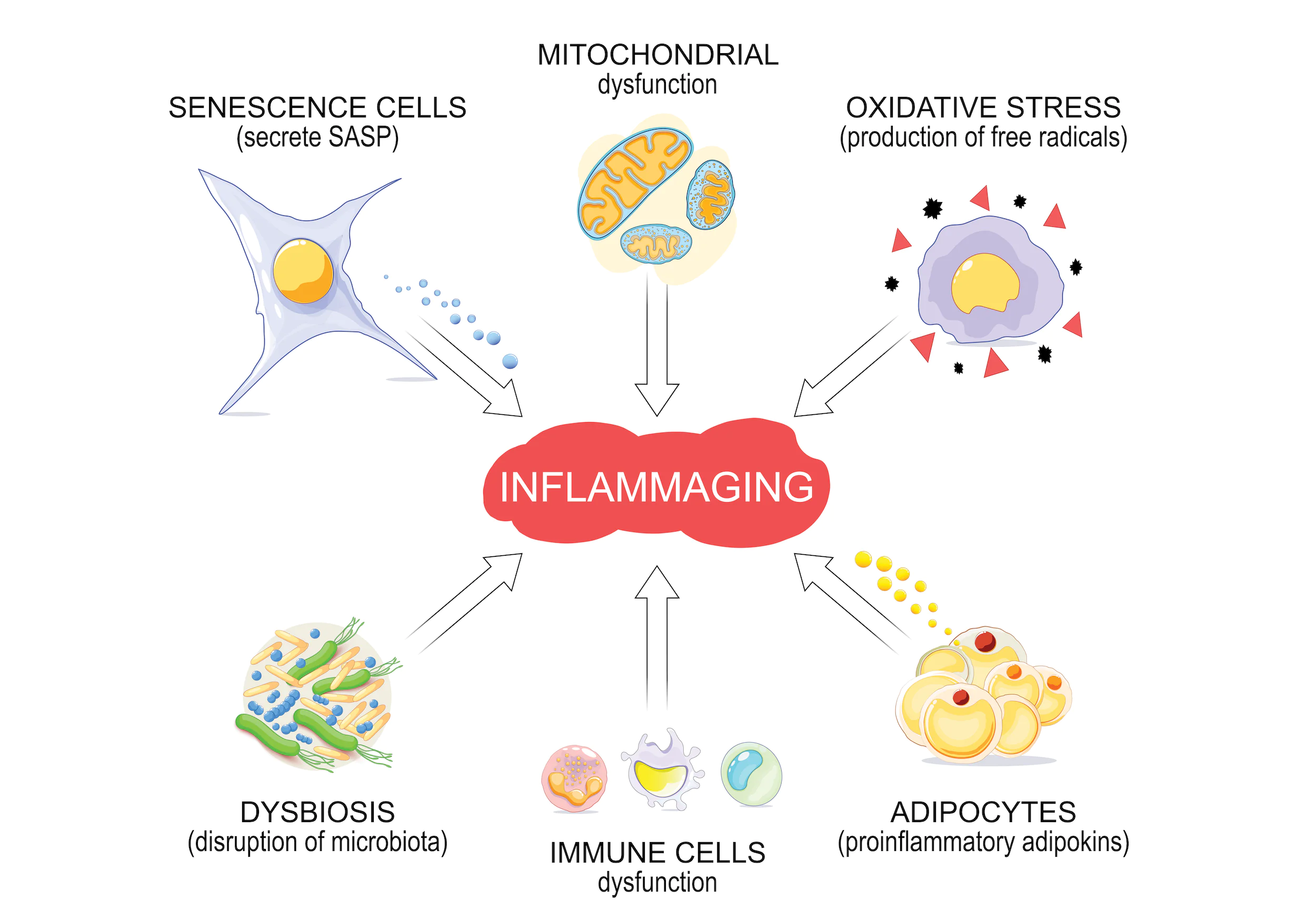
The risk for disease and co-morbidities has sky-rocketed in recent years among aging populations. This is due to numerous factors such as dysregulated innate immunity, cell senescence, prolific reactive oxygen species (ROS), accumulation of “self-debris ” or dead cell material, and microbiome dysbiosis, that both contributes to and exacerbates what we now know as inflammaging.1-3
What is Inflammaging?
Inflammaging is the chronic display of low-grade inflammation, occurring without infection or pro-inflammatory stimuli, that degrades tissue and further contributes to the acceleration of age-related conditions such as Alzheimer’s, atherosclerosis, diabetes, sarcopenia, and osteoporosis.1,2 Such inflammation is becoming more common, where in theory, it should subside immediately after the pathogen or the acute inflammatory stimuli has been addressed, allowing the tissue to rest, repair, and heal. This chronic state of inflammation, however, weakens and degrades the tissue, making it less resilient and more prone to further assault and injury, which further weakens the body systems and body overall.1
This type of chronic inflammation causes hyperactivity of the innate immune system (broad-spectrum immunity) which is already upregulated as adaptive immunity (targeted immunity) decreases naturally with age.2,3 This dysregulation impairs the body’s natural ability to efficiently and effectively respond to incoming antigens or environmental stimuli and maintain homeostatsis.3
Cytokines

An over-active immune system leads to the over-production of cytokines and other inflammatory mediators, where an abundance of cytokines weaken the anabolic signaling, or tissue building, cascade which may downregulate insulin, erythropoietin, and other hormone signaling which inhibits protein synthesis and may further attribute to sarcopenia or osteoarthristis.1,2,4 Furthermore, other pro-inflammatory cytokines have been attributed to age-related endocrine dysfunction, metabolic and digestion disruption, as well as cognitive decline, dementia, and other neurodegenerative conditions.3
Adipokines
Adipokines, or adipose-specific derived cytokines, have also been shown to trigger the proliferation of adipose tissue, which also recruits additional pro-inflammatory cytokines to these tissues. In other words, adipose tissue increases with age and accumulates in or around the liver, bones, muscles, as well as other organ tissues, and further triggers an inflammatory cascade response. This inflammatory cascade causes more cytokines to be directed to localized tissue, further promotes more accumulation of adipose in these organs, and the cycle continues, creating a now systemic pro-inflammatory environment.
Macrophages
Another inflammatory mediator, macrophages- normally involved in acute inflammation, have also been shown to be more abundant and prolific in age-related inflammation. They infiltrate the adipose tissue and, like adipokines, induce adipose accumulation, trigger adipose inflammation – or the release of adipokines, and further contribute to systemic inflammation.3 Other tissues, such as the muscles, kidney, heart, liver, and brain commonly experience elevated levels of macrophages during aging, though it is thought macrophages in these areas attribute to more of a localized pro-inflammatory response.
Microglias
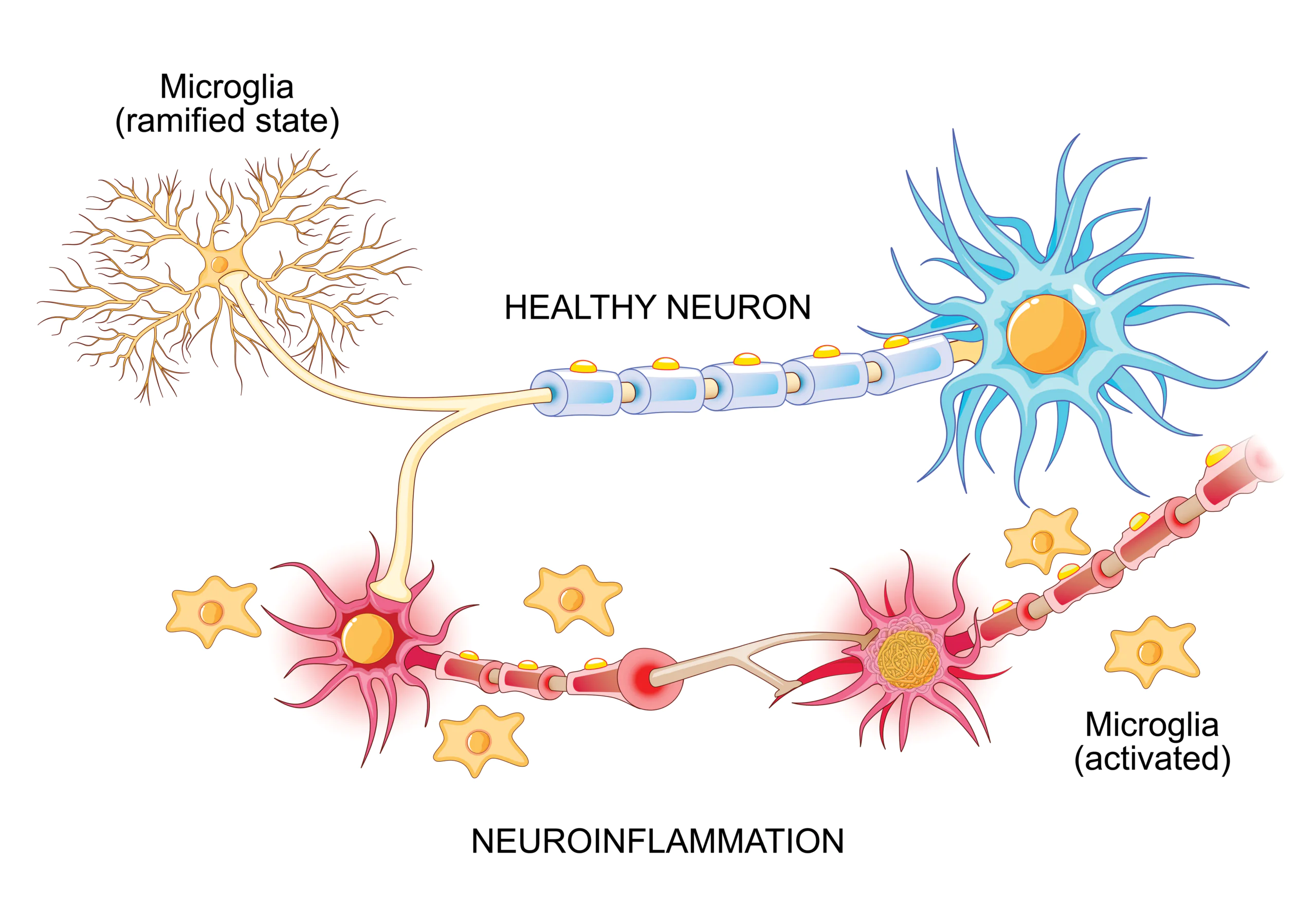
However, like cytokines, microglias, or brain-specific macrophages, trigger yet another inflammatory cascade, and have also been attributed to the onset of dementia, again, demonstrating the link between inflammaging and age-related conditions.3 Interestingly, a recent Japanese study on semi-supercenternarians, those who are between 105-109 years old, concluded that chronic systemic inflammation was a major factor in loss of cognitive function and overall mortality.
The absence of inflammation was a greater predictor of longevity more so than telomere length, the length of the repetitive sequence at the end of a DNA strand, which has been previously established as the predictor of successful aging.3 Telomere length, along with cell damage and oxidative stress, are some of the potential mechanisms attributed to cell senescence. Cell senescence, or the irreversible end of the cell-cycle where the cell is no longer able to replicate, also triggers the release of various pro-inflammatory cytokines. It is thought that this pro-inflammatory response at the end of a cell’s life is the original cause of inflammaging and the corresponding age-related conditions.1
Detoxification Pathways in Aging Populations
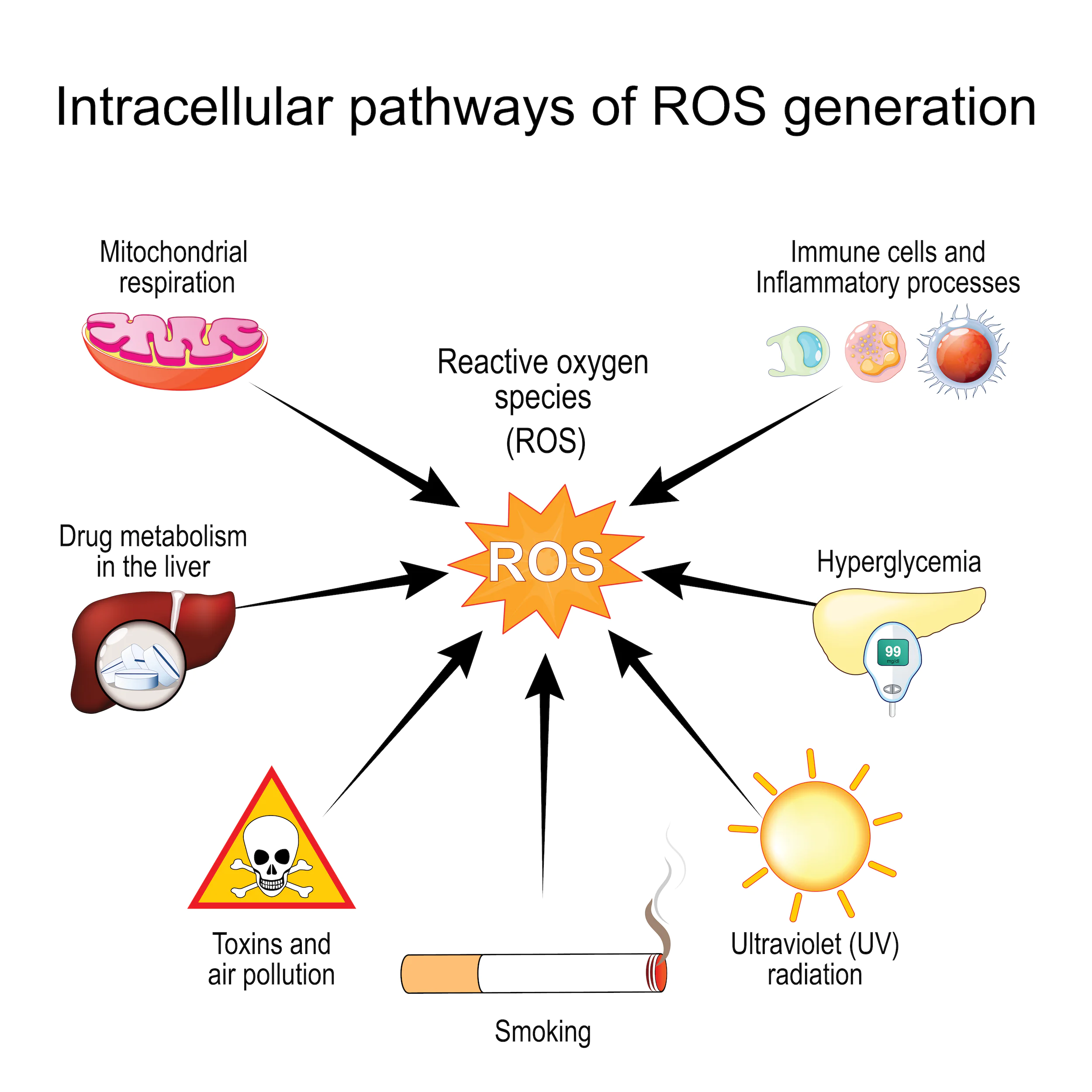
Additionally, it has been shown that the clearance of these now dead cells, slows the progression of, and may even prevent, age-related conditions, such as atherosclerosis and osteoarthritis.1 However, clearance of this dead material, or self-debris, is challenging as the number of cell deaths increase with age, as well as the pathways of clearance and elimination also decrease with age. These cellular components, such as free radicals, and metabolites, are recognized by a series of “hazard” sensors, called inflammasomes, that again trigger a pro-inflammatory response in the hopes of debris clearance and/or cellular repair.
When a cell is damaged or dies, the inflammasomes cause a release of mitochondria-specific reactive oxygen species (ROS) that initiate their own highly potent inflammatory cascade loop, causing further inflammation and damage to the cell and surrounding tissues.1 The ability for the cell to handle oxidative stress and other reactive species is well-functioning in youth but declines or malfunctions as we age, also resulting in the activation of the pro-inflammatory response system.3 As the damaged cells accumulate, the “hazard” response becomes chronic, and further triggers a maladapted, highly inflammatory response that further alters the microenvironment of the surrounding tissue.1
Gut Microbiome
Another microenvironment modulated by inflammaging is the gut microbiome. While it has been shown that the presence of Bifidobacterium is inversely correlated to serum levels of inflammatory cytokines, it has also been documented that these and other anti-inflammatory/health promoting microbiota decrease with age. To add to this, the more pro-inflammatory and pathogenic species have also been found to increase with age. These pro-inflammatory changes in gut microbiota may also increase the susceptibility of infection and pathological colonization of undesirable microbiota in this population.1 Furthermore, these detrimental microbiota produce other inflammatory byproducts and metabolites that leach into the surrounding tissue and eventually end up in circulation, causing- and exacerbating- damage along the away. In fact, this increased microbial translocation has been shown to result in endotoxemia, destruction of the vascular integrity, altered blood flow or stasis, and atherosclerosis. Of course, the body also loses the ability to control and sequester these microbes and their metabolites with age, further contributing to this dysbiosis.2
Oral Dysbiosis
It’s not just the gut microbiota that is contributing to inflammaging, the oral microbiota is also a contributing factor. Oral dysbiosis, in combination with the body’s natural degradation of the gums over time and maladjusted inflammatory response, has been shown to both cause and worsen gingivitis and periodontal disease. The presence of these conditions has been linked to a significant decline in quality of life, as well as specific age-related pathologies such as cardiovascular disease, endothelial dysfunction, metabolic dysfunction, diabetes, and neurodegenerative diseases due to the spillover of inflammatory cytokines from the periodontium into circulation. Oral dysbiosis also increases the risk of periodontal pathogens that can further impede and modulate the immune system, while creating a more favorable environment for these opportunistic microbes to thrive. Furthermore, as the periodontal tissue deteriorates over time, the inflammatory response starts to malfunction thus triggering a flood of cytokines to the localized tissue and causes yet another inflammatory cascade loop. This inflammatory cascade then triggers even more tissue degradation and cell death, exacerbating it further.5
However, there is hope. It’s not all doom and gloom as inflammaging is both preventable and curable.2
Phytonutrients in Whole Foods and Their Influence on Inflammaging
Dietary choices, good or bad, are believed to have a major influence on both the development and progression of age-related diseases. In particular, the Mediterranean diet, which is high in polyphenols, has been shown to profoundly modulate the systemic inflammatory response by inhibiting the production of such inflammatory metabolites and further protect against other pro-inflammatory damage. A diet such as this could potentially attenuate age-related diseases, improve cell metabolism and systemic outcomes, and further promote overall quality of life.6 In fact, adherence to this diet has been largely associated with decrease pain, disability, and depression as well as an increase in physical performance, cognitive function, and overall vitality.4
Whole Food Nutrition: The Mediterranean Diet

The Mediterranean diet is a well-balanced diet, characterized by consistent use of olive oil, vegetables, fruits, nuts, legumes, whole grains, and seafood as well as the moderate inclusion of eggs, dairy, and other lean proteins. It also emphasizes the importance of restricting saturated fats, red meats, processed foods, refined grains, and sugar. All in all, it provides a well-balanced mix of antioxidants and anti-inflammatory components, as well as comprehensive microbiome support such as prebiotic compounds and beneficial fiber.6,7
Polyphenols: Resveratrol, Catechins, and Flavonoids

As mentioned earlier, this diet is high in polyphenols and other phytonutrients which have developed in an evolutionary way to protect the plant from pathogens, insects, animals, etc. A similar cell-defense mechanism is imparted to us humans upon consumption of these phytonutrients.6 Some of these phytonutrients and polyphenols include resveratrol- found in grapes, catechins- found in green tea, and flavonoids, such as quercetin – found in dark berries, dark chocolate, and citrus fruits.
These compounds have been extensively studied and their numerous mechanisms of action can be distilled down to the potent and direct antioxidant activity that occurs at the intra-cellular level, as well as the inhibition of cytokine activity, the regulation and modulation of gene expression, and the activation of additional anti-inflammatory cascades. Essentially, they have far-reaching benefits and have been notably recognized for their promotion and protection of the cardiovascular system, nervous system, musculoskeletal system, immune system, and beyond.8
Clinical Takeaways

In conclusion, while the compounding factors of inflammaging such as a dysregulated immune system, self-debris proliferation, and microbiome dysbiosis are incredibly detrimental, a change in diet could potentially alleviate, prevent, and reverse this type of chronic inflammation. The Mediterranean diet, based on adequate intake of olive oil, vegetables, fruits, legumes, and seafood, is high in polyphenols and microbiome-promoting compounds, and may ease the overall process of aging and inflammation.
The compounds associated with the Mediterranean diet support cell integrity and metabolism, free-radical scavenging, and proper immune response, which further promotes healing, self-regulation, and repair of damaged tissue and organ structures. Adherence to this diet may then attenuate disease and, more importantly, improve longevity and quality of life.
Did you know WholisticMatters is powered by Standard Process? Learn more about Standard Process’ whole food-based nutrition philosophy.
1. Sanada F, Taniyama Y, Muratsu J, et al. Source of Chronic Inflammation in Aging. Frontiers in Cardiovascular Medicine. 2018 Feb 22;5doi:10.3389/fcvm.2018.00012
2. Franceschi C, Campisi J. Chronic Inflammation (Inflammaging) and Its Potential Contribution to Age-Associated Diseases. The Journals of Gerontology: Series A. 2014/06/01;69(Suppl_1)doi:10.1093/gerona/glu057
3. Chung HY, Kim DH, Lee EK, et al. Redefining Chronic Inflammation in Aging and Age-Related Diseases: Proposal of the Senoinflammation Concept. Aging and Disease. 2019 Apr 1;10(2)doi:10.14336/AD.2018.0324
4. Villagrán-Andrade KM, Núñez-Carro C, Blanco FJ, et al. Nutritional Epigenomics: Bioactive Dietary Compounds in the Epigenetic Regulation of Osteoarthritis. Pharmaceuticals 2024, Vol 17, Page 1148. 2024-08-30;17(9)doi:10.3390/ph17091148
5. Amato M, Polizzi A, Viglianisi G, Leonforte F, Mascitti M, Isola G. Impact of Periodontitis and Oral Dysbiosis Metabolites in the Modulation of Accelerating Ageing and Human Senescence. Metabolites. 2025 Jan 9;15(1)doi:10.3390/metabo15010035
6. Martucci M, Ostan R, Biondi F, et al. Mediterranean diet and inflammaging within the hormesis paradigm. Nutrition Reviews. 2017/06/01;75(6)doi:10.1093/nutrit/nux013
7. Jacquier EF, Kassis A, Marcu D, et al. Frontiers | Phytonutrients in the promotion of healthspan: a new perspective. Frontiers in Nutrition. 2024/07/12;11doi:10.3389/fnut.2024.1409339
8. vel Szic KS, Declerck K, Vidaković M, et al. From inflammaging to healthy aging by dietary lifestyle choices: is epigenetics the key to personalized nutrition? Clinical Epigenetics 2015 7:1. 2015-03-25;7(1)doi:10.1186/s13148-015-0068-2