A Synbiotic Approach to Pediatric Microbiome Health
A child with a sensitive stomach may present with frequent colds or seasonal allergies. Another with persistent eczema may struggle with digestion or focus. More often than not, these concerns do not exist in isolation. For years, these patterns were treated as separate clinical puzzles. Today, they are increasingly understood as part of a larger conversation within the body, one that often begins in the gut.
A microbiome-first framework for whole child resilience
A microbiome-first approach to pediatric health is gaining traction not as a trend, but as a reflection of emerging science. The developing microbiome, the maturing immune system, and broader communication networks throughout the body are deeply interconnected and still being shaped in real time during childhood. This period represents a window of remarkable potential. Much like a young child can effortlessly absorb new languages, the microbiome in early life is especially receptive, capable of building resilience that can influence health patterns for years to come.
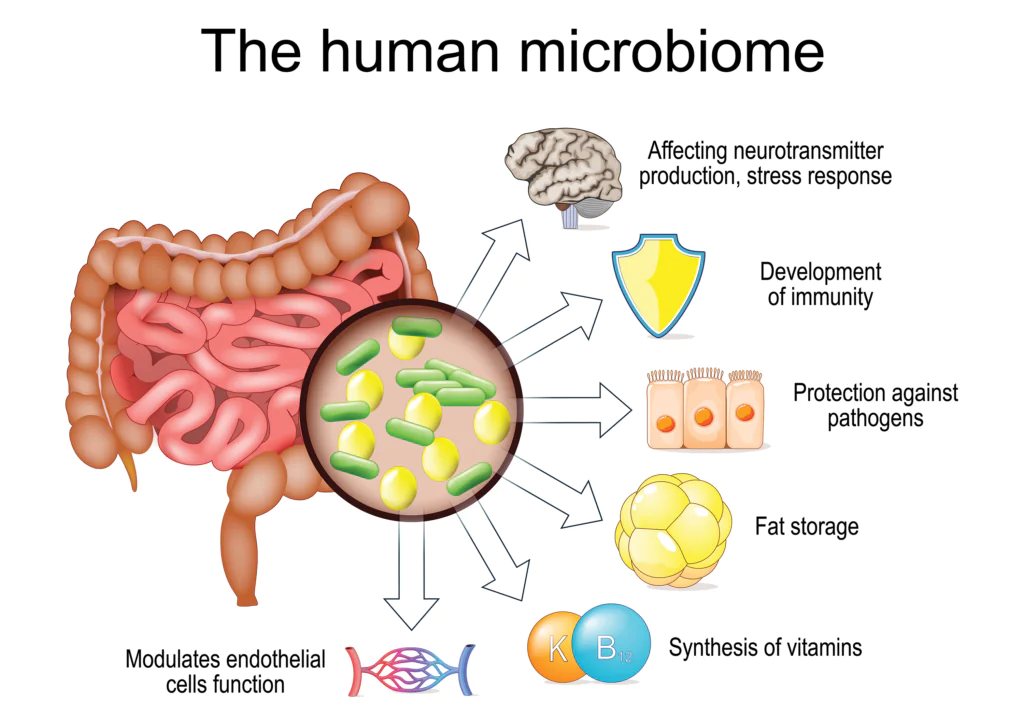
This understanding is shifting how we think about support, moving away from a paradigm of compensation toward a terrain-focused synbiotic strategy that helps shape the environment in which the microbiome can establish, communicate, and function over time. For clinicians, this offers an integrative perspective. Supporting the gut during this formative period is not simply about improving digestion. It is an opportunity to influence immune balance, neurocognition, skin health, allergic responsiveness, and broader patterns of health at a foundational level.
Pediatric nutrition: shaping the microbial language
In many ways, the developing microbiome is learning a language of its own. It communicates continuously with the brain, the immune system, and beyond, shaping these systems over time. Like any language, its fluency depends on exposure. The “vocabulary” of the microbiome is built from the signals it receives early in life. These include the foods children eat, the environments they interact with, and the microbial exposures that shape their internal ecosystems.

Among these influences, diet provides some of the most consistent and meaningful building blocks. From the earliest introduction of foods, dietary patterns begin to shape which microbes thrive and which do not. Diets centered around a wide variety of whole foods introduce a range of fibers and phytonutrients that expand the microbiome’s vocabulary and functional capacity. In contrast, dietary patterns dominated by ultra-processed foods tend to provide a narrower set of building blocks, limiting the diversity of the microbial ecosystem.
As children grow, the increasing complexity of their diet continues to guide how the microbiome develops, shaping its diversity and how it communicates with the rest of the body. For a developing gut language, a wholesome diet is not just supportive; it is instructive.
The pediatric fiber gap: missing key vocabulary
If diet is the most influential curriculum shaping the microbiome, fiber may be one of its earliest and most consistent teachers. Yet this is where many children fall short. An estimated 95 percent do not meet recommended fiber intake levels.¹ What is missing is not just a nutrient, but an essential layer of instruction during a critical window of development. Clinically, the signs are often familiar. Constipation is common, frequently tracing back to low fiber intake alongside shifts in the gut microbiota.
But fiber’s role extends far beyond bowel regularity. It helps shape the microbiome itself, supporting the gut as a central hub that influences immune function, metabolism, and signaling across multiple systems. Even short-term reductions in fiber intake have been associated with measurable shifts in the gut microbiome, highlighting how quickly this ecosystem responds to dietary change.²
Over time, the continual cycle of fiber intake, microbial fermentation, and metabolite production helps guide how the microbiome develops and functions. In this way, fiber becomes more than nourishment and actually becomes an active participant in the dynamic relationship between diet, microbes, and the host.
Short-chain fatty acids: translating microbial activity
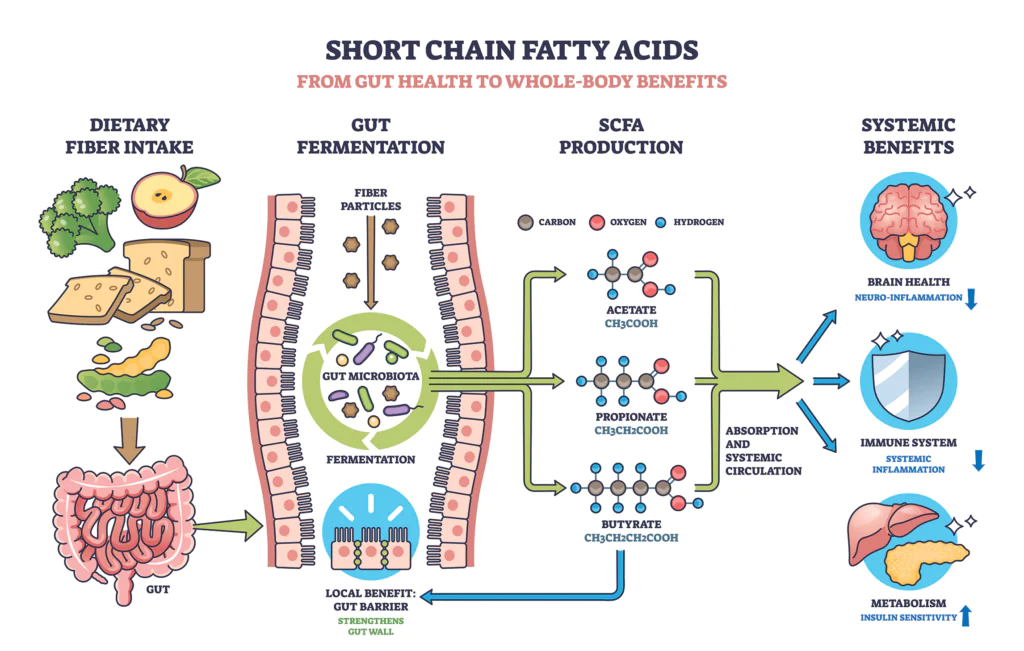
Short-chain fatty acids, or SCFAs, are important metabolites produced by the microbiome in response to dietary fiber. As fiber reaches the colon, it becomes fuel for microbial fermentation, giving rise to compounds such as acetate, propionate, and butyrate. These metabolites help regulate immune responses, support gut barrier integrity, and influence metabolic and neurological signaling throughout the body, essentially translating microbial activity into physiological effects.3
Because SCFA production depends on the availability and type of fermentable fibers, they offer a clear link between what we eat, the health of the gut lining, and how the microbiome communicates with the rest of the body. This naturally leads to an important question in practice: how can we more intentionally support the microbial processes that give rise to these metabolites?
Prebiotic fibers: fueling the conversation
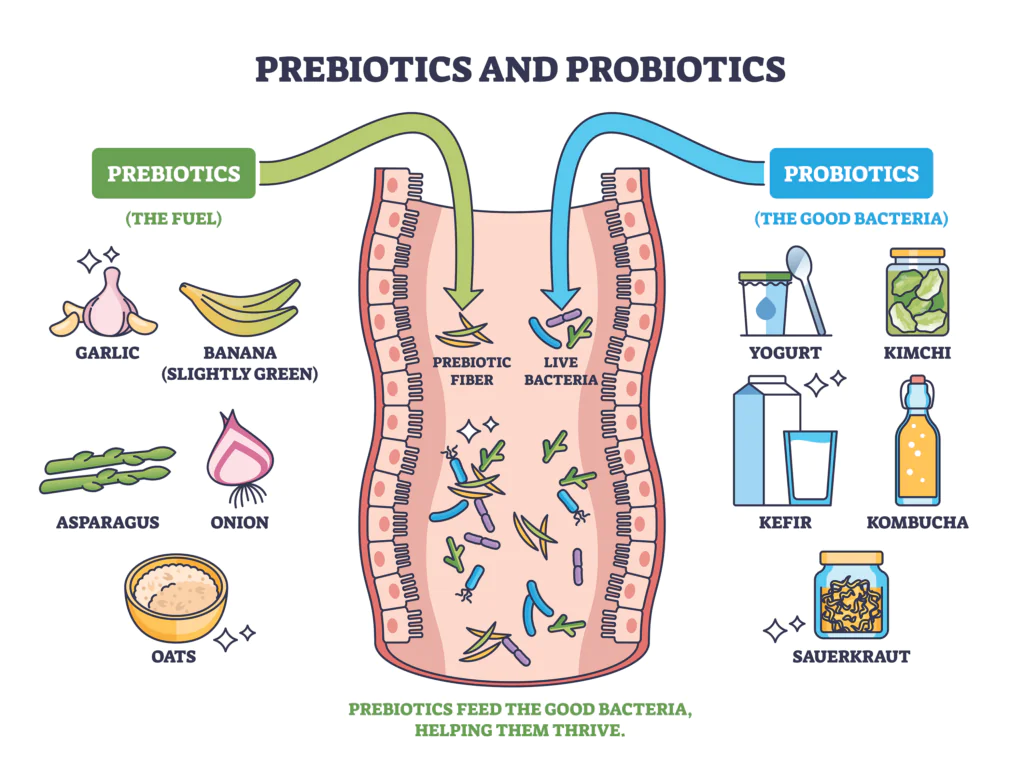
Certain fibers have a unique ability to nourish beneficial microbes and support the processes that give rise to SCFAs. These are known as prebiotic fibers. Because they pass through the upper digestive tract intact, prebiotic fibers arrive in the colon ready to be used as a reliable fuel source for specific microbial communities. As these microbes metabolize prebiotic fibers through fermentation, they contribute to the production of short-chain fatty acids and help support a more balanced microbial environment that influences immune, metabolic, and gut barrier function.
Some of the most well studied of these fibers are inulin and oligofructose. They are naturally found in foods that have long been part of the human diet, including chicory root, a hardy plant that grows in open fields and has been used for generations. Chicory root has traditionally been roasted and brewed as a coffee alternative, valued not only for its rich, bitter flavor but also for its gentle support of digestion.
Today, prebiotic fiber is recognized for its ability to nourish microbiomes in more targeted ways. Its effect is not only theoretical. In young children, even short periods of supplementation have been shown to shift the microbiota in favorable ways while improving digestive comfort. In one study of children aged 7 to 19 months, three weeks of prebiotic fiber intake was associated with fewer episodes of diarrhea, vomiting, flatulence, and fever.4
Fiber diversity: building a richer vocabulary
For growing children, the goal is not simply more fiber, but fiber from a variety of whole foods. Each type of fiber delivers a different substrate to the microbiome, helping expand its functional capacity over time. Many familiar foods provide these distinct fibers in ways that are both practical and accessible for families:

Pectin: soft support
Found in apples and other fruits, pectin forms soft, hydrating gels in the digestive tract. It supports comfortable stool formation while also serving as a fermentable fuel source for beneficial microbes.

Beta glucans: bridging systems
Present in oats, beta glucans are well studied for their role in metabolic health. They also interact with immune pathways in the gut while providing fermentable support for the microbiome.

Resistant starch: reaching deeper conversations
Found in foods such as green banana flour, resistant starch resists digestion in the small intestine and reaches the colon intact, where it fuels microbial fermentation and supports the production of butyrate, a short-chain fatty acid that fuels colonocytes, strengthens intestinal barrier integrity, and modulates inflammatory signaling.

Whole plant fibers: layered communication
Vegetables such as beetroot provide a natural mix of fibers that support both motility and microbial fermentation, allowing for more complex and sustained microbial interactions.
These fibers span both soluble and insoluble types, each contributing in different ways. Some help form softer, more hydrated stools, while others add bulk and support regular movement through the digestive tract.
Probiotics: restoring harmony
If fiber helps shape the environment, well-characterized probiotics can help bring the microbiome back into balance. Rather than introducing something foreign, they support organisms that are already familiar to the system, helping restore coordination within the microbial community.

Many of the organisms used in pediatric care reflect this idea. Species such as Lactobacillus and Bifidobacterium are common early colonizers and remain central to a well-balanced microbiome throughout childhood. Supporting these organisms is less about adding new voices and more about strengthening those that help the system stay in tune.
At the same time, clinical outcomes are not defined at the level of species alone. Probiotic effects are determined at the strain level, and much of the evidence in pediatric care is tied to well-characterized strains studied for specific outcomes. While parents may recognize familiar species names, it is the individual strain that determines how a probiotic behaves within the body.
In pediatric digestive health, strains such as Lactobacillus acidophilus DDS-1 and Bifidobacterium lactis UABLA-12 have been studied extensively for their role in supporting bowel function, with improvements in stool frequency, consistency, and overall bowel habits in children with functional constipation.5
These organisms also play an important metabolic role. Both are lactate producers, contributing to a pool of metabolites that can be used by other beneficial microbes. Through this process, often referred to as cross-feeding, they help support the growth and activity of additional microbial communities involved in short-chain fatty acid production and overall gut stability. In this way, probiotics do more than act alone. They help lay the metabolic groundwork for a broader microbial network. But these interactions depend on having the right substrate. Without adequate fiber, the system has little to build on.
Synbiotics: creating coherence
When probiotic microbes are paired with supportive fibers, they form a synbiotic, bringing together beneficial organisms with the nutrients that nourish them. Rather than introducing microbes in isolation, synbiotics help create an environment where microbial activity is more coordinated and better sustained.
In children, this synbiotic strategy makes sense. The pediatric microbiome is not simply maintained. It is being built. To build something resilient, the terrain must be supportive. The inputs provided during this time influence not only which microbes are present, but how effectively they communicate with the immune system, the skin, and other systems throughout the body.
Immune tone: clinical outcomes with synbiotics
In clinical research, this integrated approach shows meaningful outcomes, particularly in conditions that reflect the evolving relationship between the gut and the immune system. In early childhood, this relationship is often visible through patterns such as the atopic march, where eczema, allergies, and respiratory conditions unfold along a shared immunological trajectory. Intervening during this window offers an opportunity not only to manage symptoms, but to influence the direction of that progression.
In children with atopic dermatitis, a synbiotic combination of L. acidophilus DDS-1, B. lactis UABLA-12, and inulin improved eczema symptoms across multiple domains, including the extent of skin involvement, itch intensity, and sleep disruption.6 These changes reflect more than surface-level improvement. The skin, as an outward expression of immune tone, often mirrors deeper coordination within the gut–immune axis. Shifts in CD4 to CD8 ratios observed in the study point to a softening of immune reactivity, a movement away from a more hypersensitized state. When this kind of shift begins to take place, it raises a broader question. If the immune terrain is becoming less reactive at the level of the skin, how might that influence other systems shaped by immune responsiveness, including the respiratory tract?
It certainly seems to make a difference. In children experiencing acute respiratory infections, the same synbiotic combination supported faster recovery and milder illness.7 This translated into earlier symptom resolution, improved day to day comfort, and fewer missed days from school or childcare.
A probiotic yeast: holding the line during disruption
But even in a well-supported system, disruptions still occur. Antibiotic use is a common and often necessary part of pediatric care, but it can temporarily interfere with microbial balance and contribute to gastrointestinal symptoms, including antibiotic-associated diarrhea.
One microorganism used to support digestive stability during antibiotic exposure is Saccharomyces boulardii, a probiotic yeast. Unlike bacterial probiotics, this gut familiar yeast is not affected by antibacterial medications and can remain active during antibiotic therapy where it lends a protective effect. Clinical studies show that S. boulardii helps reduce the risk of antibiotic-associated diarrhea in children and supports recovery during acute diarrhea from other causes.⁸
Within a broader synbiotic strategy that includes supportive fibers and well-studied bacterial probiotics, S. boulardii provides an additional layer of support for maintaining microbial balance and digestive resilience during periods of disruption.
Clinical Takeaway: Building microbial fluency over time
The pediatric microbiome is not fixed. It is learned, developing through exposure, repetition, and interaction, much like language. Within this process, different classes of organisms take on complementary roles. Bacterial strains such as L. acidophilus DDS-1 and B. Lactis UABL-12 support fermentation and cross-feeding, while Saccharomyces boulardii helps maintain stability during periods of disruption.
Gut microbes do not act in isolation. Their function is shaped by the environment they inhabit. A diverse fiber landscape provides the substrate for microbial metabolism, signaling, and coordination, helping guide how the microbiome develops and communicates over time.

Together, these elements begin to form a synbiotic approach, one that supports a microbiome that is not only present, but actively engaged and functional. For clinicians, this offers a meaningful opportunity to support digestive function, immune resilience, and broader patterns of health during a critical window of development. When we support a gut-first approach during childhood, we are not simply managing symptoms, but helping set the stage for a lifetime of microbial fluency.
Did you know WholisticMatters is powered by Standard Process? Learn more about Standard Process’ whole food-based nutrition philosophy.
1. Quagliani D, Felt-Gunderson P. Closing America’s fiber intake gap. American Journal of Lifestyle Medicine. 2017;11(1):80–85. doi:10.1177/1559827615588079
2. David LA, Maurice CF, Carmody RN, et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature. 2014;505:559–563. doi:10.1038/nature12820
3. Hsu CY, Chou YY, Chen YC, et al. Microbiota-derived short-chain fatty acids in pediatric health and diseases: from gut development to neuroprotection. Frontiers in Microbiology. 2024;15:1456793. doi:10.3389/fmicb.2024.1456793
4. Waligora-Dupriet AJ, Campeotto F, Nicolis I, et al. Oligofructose supplementation in young children during diarrhea: effects on intestinal microbiota and digestive health. International Journal of Food Microbiology. 2007;113(2):239–246. doi:10.1016/j.ijfoodmicro.2006.07.009
5. Gan D, Chen J, Tang X, et al. Impact of a probiotic chewable tablet on stool habits and microbial profile in children with functional constipation: a randomized controlled clinical trial. Frontiers in Microbiology. 2022;13:985308. doi:10.3389/fmicb.2022.985308
6. Gerasimov SV, Vasjuta VV, Myhovych OO, Bondarchuk LI. Probiotic supplement reduces atopic dermatitis in preschool children: a randomized, double-blind, placebo-controlled trial. American Journal of Clinical Dermatology. 2010;11(5):351–361.
7. Gerasimov SV, et al. Role of short-term use of Lactobacillus acidophilus DDS-1 and Bifidobacterium lactis UABLA-12 in acute respiratory infections in children: a randomized controlled trial. European Journal of Clinical Nutrition. 2016;70(4):463–469. doi:10.1038/ejcn.2015.171
8. Kotowska M, Albrecht P, Szajewska H. Saccharomyces boulardii in the prevention of antibiotic-associated diarrhoea in children: a randomized double-blind placebo-controlled trial. Alimentary Pharmacology & Therapeutics. 2005;21(5):583–590. doi:10.1111/j.1365-2036.2005.02356.x