Part 9: Immune System Series | The Role of Aging, Exercise, and Diet in Immune Function
Summary
Exercise and lifestyle throughout life are vital to maintain health throughout the life span, and may reduce the risk of chronic disease.
Aging significantly affects social and financial resources. By 2050, 20 percent of the global population will be 60 years of age or older.1 Living longer is a double edge-sword, as it brings healthy aging to some and debilitating chronic degenerative diseases to others.
How Can Lifestyle Choices Improve Health?
Exercise and lifestyle throughout life are vital to maintain health during all stages of life, and may reduce the risk of chronic disease.2 However, older individuals have a reduced capacity to induce muscle hypertrophy with resistance exercise, which can lead to sarcopenia.3 Interestingly, a study of older versus younger men demonstrated a reduced adaptability of aging muscle to exercise, as demonstrated by the absence of microRNA (miRNA) regulation and blunted transcription of mRNAs. There is a mechanistic role of miRNA in the adaptation of muscle to anabolic stimulation, and it is clear that there is an impairment in exercise-induced miRNA/mRNA regulation with aging.4
Nutrients in the diet are powerful tools to modulate aging; however, nutrient sensing pathways deregulate and lose effectiveness with age.5 mRNAs have emerged as important regulators of cellular function and can be modified by the diet. Some mRNAs target genes encoding proteins and enzymes, belonging to nutrient sensing pathways and may play a major role in the modulation of the aging process. Some nutrient sensing pathways impaired by aging play key roles in the regulation of protein synthesis, cell cycle, DNA replication, autophagy, stress responses, and glucose homeostasis.
What Biological Hallmarks Contribute to Aging?
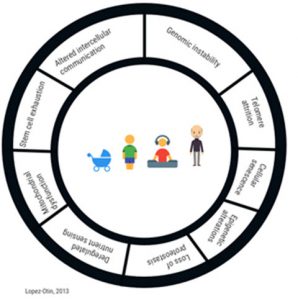
There are many biological hallmarks that contribute to aging:6
- Genetic and epigenetic factors
- Telomere shortening
- Proteostasis
- Impaired signaling pathways
- Reduced stem cell pluripotency
- Cellular senescence
- Mitochondrial and immune dysfunction
- Alterations in nutrient sensing pathways
In addition, a prominent alteration in intercellular communication with aging is “inflammaging.” This is a term describing the smoldering proinflammatory phenotype that accompanies the process of getting older. Inflammaging likely has many causes, but at the core of the problem is a failure of the inter-connected immune system (innate and acquired) to effectively resolve the inflammation and return to homeostasis.
How Can Clinicians Support Healthy Aging?
Given all of the challenges of inflammation associated with aging and infection, what should the clinician do to support healthy aging and reduce chronic inflammatory conditions? Typically, clinicians will prescribe non-steroidal anti-inflammatory drugs to immuno-suppress (block or inhibit) the inflammation pathway. However, use of anti-inflammatory agents may actually prolong the inflammatory tone and cause unwanted side effects, such as chronic inflammation.7
In addition to healthy diet and lifestyle, there is now an emerging strategy to support the natural pro-resolution pathway via a super-family of chemical mediators that stimulate the resolution of the response, known as specialized pro-resolving mediators (SPMs). SPMs are intracellular enzymatic oxidation derivatives of long chain n-3 fatty acids (eicosapentaenoic acid, EPA 20:5n3 and docosahexaenoic acid, DHA 22:6n3), as well as long chain n-6 derived intermediates of arachidonic acid, (AA 20:4n6). Collectively, SPMs, derived from the diet, and also called resolvins, are physiologic mediators and pharmacologic agonists that stimulate resolution of inflammation and infection.8-12
Additionally, one type of SPM, resolvin D1 (RVD1) has been shown to engage an RvD1 receptor-dependent regulation of specific miRNA involved in the inflammatory response. RvD1 targets resolvin-regulated miRNAs involved in the inflammatory cascade such as miR-146b (NFκB signaling) and miR-219 (targeted 5 lipoxygenase), resulting in reduced leukotriene production. Thus, clinicians may recommend n-3 fatty acids to enrich tissues with EPA and DHA to increase membrane fluidity and to support the resolution of inflammation with resolvins.
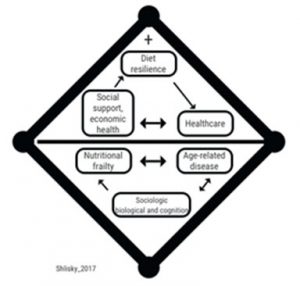
A healthy diet and a lifestyle that involves regular exercise both continue to be excellent clinical management tools for clinicians that are managing patients with inflammation. The importance of diet is nicely illustrated in animal trials, in which vitamin E supplementation reduced atherosclerosis and mortality, but not if the animals were consuming a Western style diet.13 Not only can excellent nutritional quality reduce the incidence of age-related disease, it can also improve the prognosis for age-related disease. Factors that positively and negatively influence nutritional health are nicely illustrated in the Figure adapted from Shlisky et al.14 Adults older than 60 years have increased incidence of infectious and inflammatory diseases with prolonged recovery time, higher morbidity, and higher mortality from these diseases. Researchers largely attribute disease onset to immune dysregulation in which the cell-mediated immune response is compromised in part through reduced nutritional status and bioactive dietary components: polyphenols, omega-3 long chain fatty acids, and others.
An important consideration when evaluating the elderly is the degree of nutritional quality and consumption of nutrient-rich foods. The elderly may have a reduced capacity to absorb and utilize essential nutrients than in their younger years, even without dietary changes. Thus, supplementation of essential nutrients and bioactive components, including bioactive fatty acids, may enhance the function of the immune system and may be an effective strategy to reduce the morbidity and mortality associated with age-related diseases.14-16
Read part 10 of the Immune System Series: Implications for Clinicians in Support of a Healthy Immune System.
- Cannata, A., Marcon, G., et al. (2017). Role of circulating factors in cardiac aging. J Thorac Dis 9(Suppl 1): S17-S29.
- Russell, A. P., Lamon, S. (2015). Exercise, skeletal muscle and circulating microRNAs. Prog Mol Biol Transl Sci 135: 471-496.
- McGregor, R. A., Poppitt, S.D., et al. (2014). Role of microRNAs in the age-related changes in skeletal muscle and diet or exercise interventions to promote healthy aging in humans. Ageing research reviews 17: 25-33.
- Rivas, D. A., Lessard, S.J., et al. (2014). Diminished skeletal muscle microRNA expression with aging is associated with attenuated muscle plasticity and inhibition of IGF-1 signaling. FASEB journal : official publication of the Federation of American Societies for Experimental Biology 28(9): 4133-4147.
- Mico, V., Berninches, L., et al. (2017). NutrimiRAging: micromanaging nutrient sensing pathways through nutrition to promote healthy aging. International journal of molecular sciences 18(5).
- Lopez-Otin, C., Blasco, M.A., et al. (2013). The hallmarks of aging. Cell 153(6): 1194-1217.
- Serhan, C. N. (2017). Discovery of specialized pro-resolving mediators marks the dawn of resolution physiology and pharmacology. Mol Aspects Med Article in Press.
- Wang, C. W., Colas, R.A., et al. (2016). Maresin 1 biosynthesis and proresolving anti-infective functions with human-localized aggressive periodontitis leukocytes. Infection and immunity 84(3): 658-665.
- Whelan, J., Gowdy, K.M., et al. (2016). N-3 polyunsaturated fatty acids modulate B cell activity in pre-clinical models: Implications for the immune response to infections. European Journal of Pharmacology 785: 10-17.
- Zhu, M., Wang, X., et al. (2016). Pro-resolving lipid mediators improve neuronal survival and increase abeta42 phagocytosis. Molecular neurobiology 53(4): 2733-2749.
- Chiang, N., de la Rosa, X., et al. (2017). Novel resolvin D2 receptor axis in infectious inflammation. The Journal of Immunology 198(2): 842-851.
- Perretti, M., Cooper, D., et al. (2017). Immune resolution mechanisms in inflammatory arthritis. Nat Rev Rheumatol advance online publication.
- Meydani, M., Kwan, P., et al. (2014). Long-term vitamin E supplementation reduces atherosclerosis and mortality in Ldlr-/- mice, but not when fed Western style diet. Atherosclerosis 233(1): 196-205.
- Shlisky, J., Bloom, D.E., et al. (2017). Nutritional considerations for healthy aging and reduction in age-related chronic disease. Adv Nutr 8(1): 17-26.
- Echeverri Tirado, L. C., Yassin, L.M. (2017). B cells interactions in lipid immune responses: implications in. Lipids in health and disease 16(1): 1-11.
- Nadjar, A., Leyrolle, Q., et al. (2017). Bioactive lipids as new class of microglial modulators: When nutrition meets neuroimunolog. Progress in Neuropsychopharmacology & Biological Psychiatry 79: 19-26.